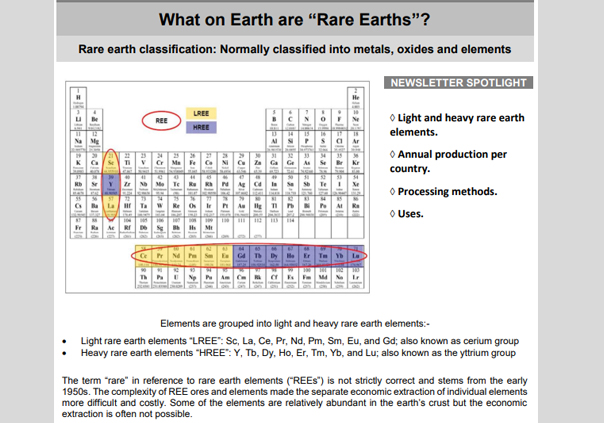
The International Union of Pure and Applied Chemistry defines REEs as the 15 Lanthanide elements plus Scandium and Yttrium. These 15 elements share common physiochemical properties. Due to this these Lanthanides often occur together as elemental constituents of their host minerals. Two other metals commonly found in association with lanthanides in the same minerals and sharing similar physiochemical properties are Scandium (21 Sc) and Yttrium (39 Y).
REEs do not occur as native elemental metals in nature, only as part of the chemistry of host minerals. Although there are more than 200 known REE-bearing minerals, only three are considered to be the principle REE mineral ores most fitted to the extraction of REEs, namely Bastnasite, Xenotime and Monazite.
A carbonatite orebody in Mountain Pass California USA is a large producer of REEs but is currently not producing.


The ores can be mined from beach placers such as heavy mineral sands or by open cast mining using mechanical shovels or draglines. The ore is transported to a mill where it is crushed and milled, followed by gravity concentration of the heavy minerals, and electromagnetic and high-intensity electrostatic separation. Flotation is also used for some of the minerals.
At Mountain Pass, USA, the ore is crushed and milled. The REEs are then floated off as a concentrate of about 63% REE purity. Leaching the concentrate with HCL to remove the calcite and finally roasting, increases the purity to ~92% REE.
Processing
The processing and recovery of individual REE elements or oxides is a complex series of individual chemical processes. Monazite concentrates can be treated using sodium hydroxide digestion or acid digestion.
The individual REEs can partly be separated using classical precipitation and filtration techniques. The final separation of the individual REEs is done using solvent extraction using ion selective reagents and further refining to pure individual metals or oxides as required by the market. Rare earth metals are produced most successfully by electrolysis or by metallothermic reduction of rare earth halides.
Malaysia has the largest REE refinery at present but others are in the process of design piloting and construction. China also has refining knowhow but information on the processes utilised are difficult to find.
What are REEs used for?
Although REEs have been used since the 1950s, some REEs were only discovered more recently. The use of the individual REE and the metals and oxides have found their way into electronic devices and elements used in modern day technology. Some of these applications can be considered strategic in nature.

Rare earths are not categorically rare, but are finding new uses in the modern technological world. As the demand for specific REEs increases, so too will prices increase. The production of the REEs will follow the economic balance of supply and demand but the control from China will make the price forecasts difficult to predict. South Africa has significant resources at Zankopsdrift and Steenkampskraal which can be developed successfully once the prices reach a favourable level to draw interest from international investors.