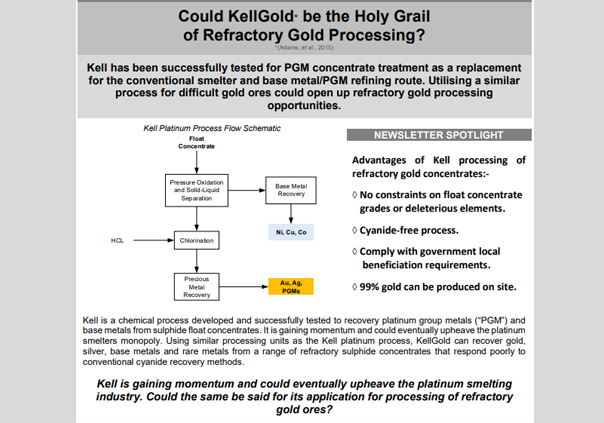
KellGold utilises commercially proven unit operations and learnings from processing of PGM concentrates through the Kell Process. The following benefits could be realised for KellGold (Adams, et al., 2015):-
• No constraints on flotation concentrate grades or impurities which could increase float mass pull and recoveries;
• Cyanide free gold recovery which negates the need to destroy the cyanide in tailings streams – some countries ban the use of cyanide altogether;
• Reduced water usage, carbon dioxide emissions and electricity usage;
• Improved profitability due to higher metals recoveries and lower operating costs in comparison with toll smelting (this would have more of a benefit for gold producers in remote countries that do not have nearby gold refineries);
• Ability to produce refined metals at the mine site which would comply with certain governments beneficiation requirements.
Toll refining in South Africa (at Rand Refinery) costs less than 1% of the gold produced. As a result, smelting costs savings would be more prevalent for gold producers in countries that do not have a gold refinery. Reagent consumer metals such as nickel, copper and cobalt are first selectively removed through a pressure oxidation step. A chloride leach similar to the Minataur Process (Scott and Matchett, 2005) then follows. Gold is selectively extracted from the gold-chloride species by means of solvent extraction and then scrubbing stages to remove impurities. Preliminary assessments for KellGold process (Adams et al., 2015) achieved gold (Au) recoveries of between 87% and 100% for various refractory gold-bearing ores. Conventional cyanide leaching of the same ores exhibited Au recoveries of 3% to 82%. Beyond that, no further testwork or feasibility work has been published in the public domain. This begs the question: is KellGold the answer to refractory gold processing? Referring to the figure below, various processing options are compared for a southern Africa refractory gold orebody studied by Minxcon.

The graph highlights the capital, operating cost and recovery differences between processing options considered in the project. Conventional leaching and fine grinding options were expected to achieve recoveries of 40% and 45% respectively compared with the 88%+ recoveries expected from POX and biological oxidation. Operating and capital costs for the pressure oxidation are higher than biological oxidation. Furthermore, there is a recovery benefit with pressure oxidation. Owing to the lower capital costs, availability of specialised operational expertise and its successful application in southern Africa, biological oxidation was selected as the preferred processing route for the project.
Although not considered for this refractory gold project, KellGold could offer further value to the project as it is expected to achieve a higher recovery than POX and biological oxidation with less stringent deleterious and gold grade requirements on the float concentrate. However, it is anticipated that the KellGold Process could be more capital intensive but have a similar operating cost as a biological oxidation process. With a long life of mine, a slightly reduced operating cost and improved recovery would typically outweigh the high capital.
KellGold has the potential to upheave refractory gold processing and open up further mining opportunities for orebodies that have not been economically extractable with current technologies. However, thorough amenability testwork will still be required as part of the trade-off phases of a pre-feasibility study to compare all processing methods and to make an informed decision.
References
• Adams, M.D., Liddell, K.S., Smith, L.A., 2015. The KellGold hydrometallurgical process for cyanidefree extraction of gold from refractory concentrates and feedstocks – a preliminary assessment. The Southern African Institute of Mining and Metallurgy, World Gold Conference 2015.
• Scott, S.A., Matchett, K., 2004. Minataur™: the Mintek alternative technology to gold refining. The Journal of The South African Institute of Mining and Metallurgy, 339-344.